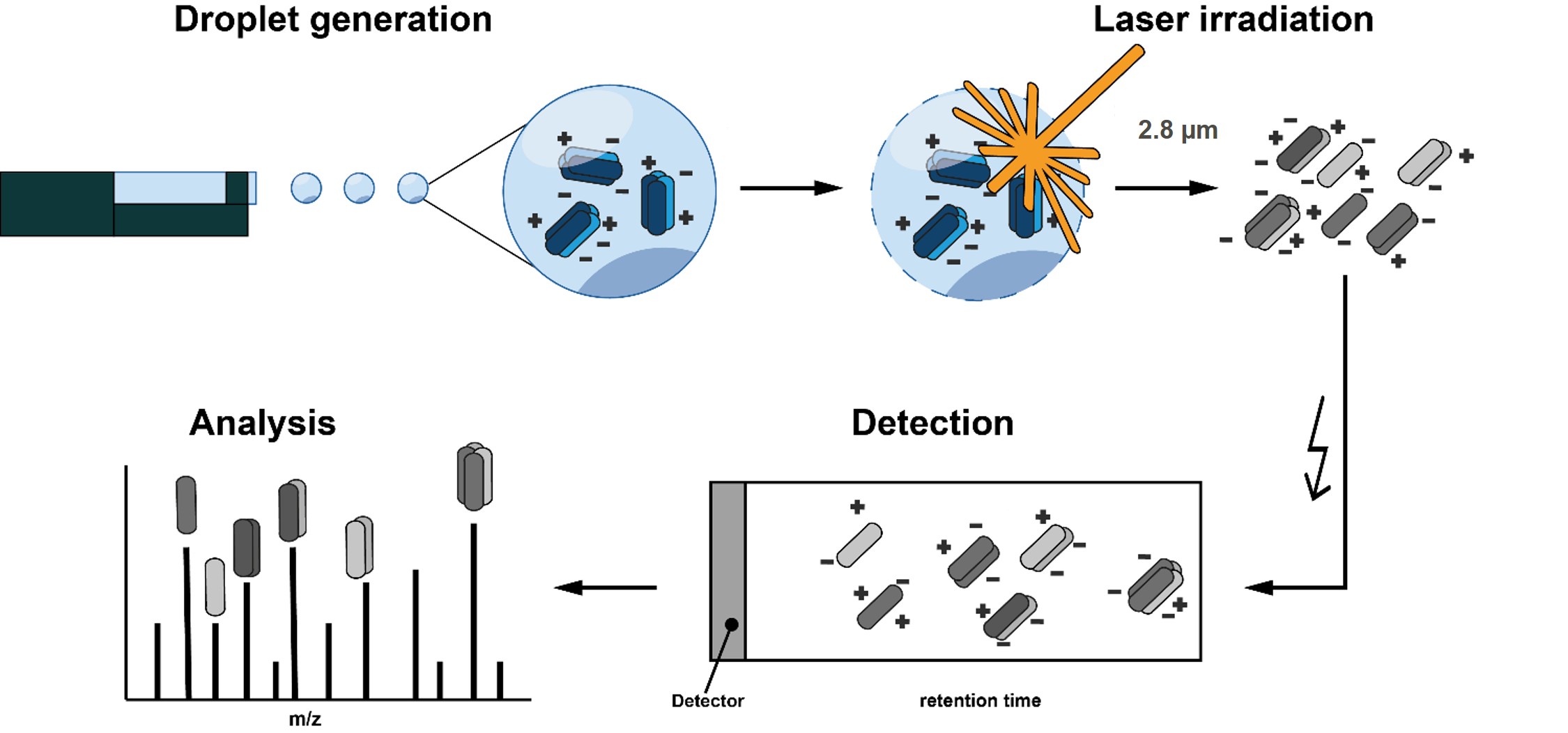
Microdroplets
(50 μm diameter) of the
sample solution are introduced into vacuum with the help of
an on
demand
droplet generator.
There they are irradiated one by one by a
mid-IR laser which runs at the same
frequency (10Hz).
The laser wave-length is tuned to the absorption maximum of water at
around 2.8 μm
- the H-O stretching vibration of water. The absorbed
laser energy leads to the explosion of the droplet, releasing the
biomolecular complex ions into vacuum, where they can be analysed by a
time-of-flight mass analyser. The incomplete ion neutralisation process
leads to gas phase ions whose charge state mimic the charge states of
the complexes in solution.
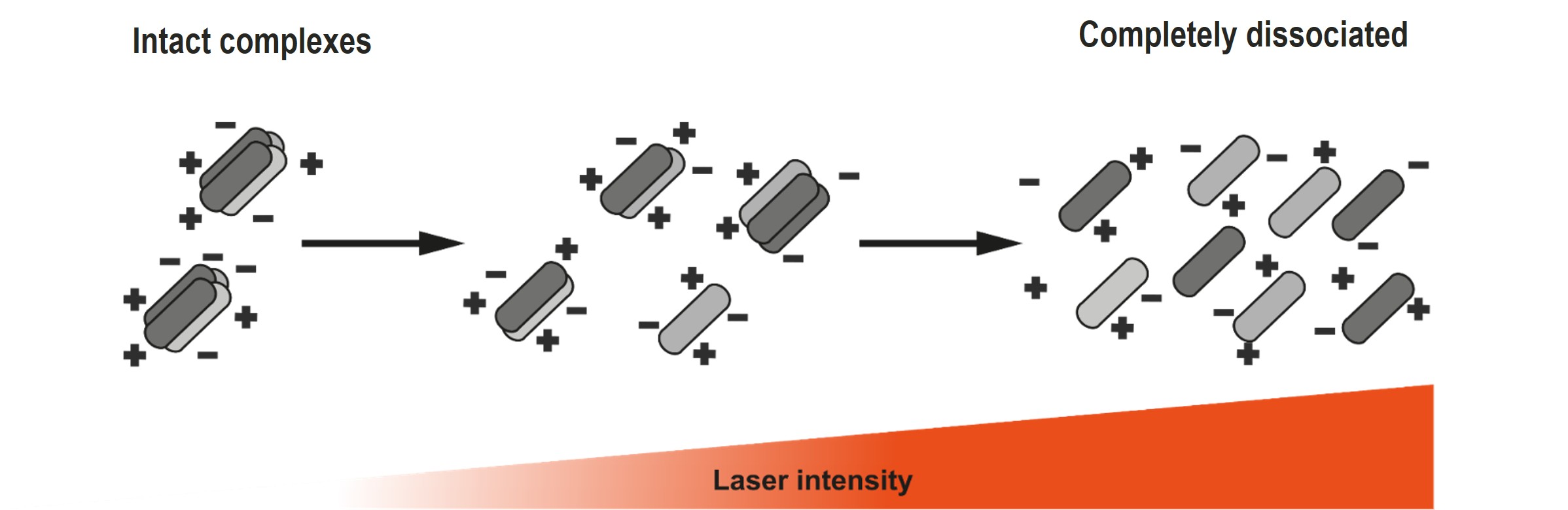
Variation of one parameter - the intensity of the desorption laser - determines the harshness of the method. Under soft conditions the complexes stay intact, while at harsh laser conditions the non-covalent bonds break, revealing the constituting subunits. At medium conditions stronger bonds can stay intact, exposing stable subcomplexes.